|
11/27/2023 0 Comments Atomic size trend anionSo we really compare apples and oranges here rather than a fairer assessment comparing say fluoride anion, and lithium cation. The pK a of the thiol group on the cysteine side chain, for example, is approximately 8.3, while the pK a for the hydroxyl on the serine side chain is on the. More importantly to the study of biological organic chemistry, this trend tells us that thiols are more acidic than alcohols. 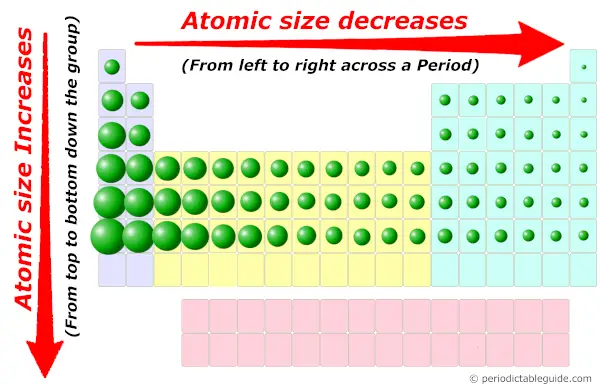
biological recognition - some ions can pass through certain membrane channels, others may be too large. Atomic Radius Versus Atomic Number 50 0 10 20 30 Atomic number 40 50 60 100 Atomic radius (pm) 150 200 250 300 Sc Zn Cd Period 1 Period 2 Period 3 Period 4 Period 5 He Ne Li Na K Rb Cs Kr Xe Ar Figure 6.14 This graph plots. Sizes of ions influence: packing of ions in ionic lattices, and therefore, the lattice energy. Figure 6.15 summa-rizes the group and period trends in atomic size. 
With respect to your initial question (I finally got there even if I did go off on a tangent!), we compare a 2nd Period non-metal, with a 3rd Period metal.and the Periodic change should be significant with regard to ionic and atomic size. HI, with a pK a of about -9, is one the strongest acids known. closer to the nucleus and the atomic size decreases. On the other hand, when an atom is REDUCED.an electron(s) is ADDED to the valence shell, and increased electronic shielding means that the resultant anion is LARGER with respect to the parent atom.Īs always, metals are electron-rich species, and TEND to be oxidized.whereas non-metals, high #Z# from the right of the Periodic Table as we face it., TEND to be REDUCED to form anions. 
But if we remove that valence electron, then inevitably the radius of the CATION should substantially decrease given that we have removed the shielding effect of an electron, and the nuclear charge contracts the radius of the remaining electrons.Īnd this effect is CERTAINLY illustrated by the given data look at the cationic radii with respect to the parent atomic radii (data are in #"1 pm"-=1xx10^-12*m#. The sizes of cations and anions follow similar trends to those of neutral atoms. (as we move from above to below in a group ) Atomic size decreases as we move from left to right in a period. (Credit: Christopher Auyeung Source: CK-12 Foundation License: CC BY-NC 3. (b) The metallic atomic radius, rmet, is half the distance between the nuclei of two adjacent atoms in a pure solid metal, such as aluminum. 
2: The atomic radius (r) ( r) of an atom can be defined as one half the distance (d) ( d) between two nuclei in a diatomic molecule. (a) The covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as Cl 2. When O gains two electrons to form O2 the. Answer: Atomic size increases with increase in period number. The atomic radius is defined as one-half the distance between the nuclei of identical atoms that are bonded together. For example if Na loses one electron to form N a+, the latter is smaller than the Na atom. Figure 2: Atomic and Ionic Radii of Some Elements. Gaining electrons increases the apparent diameter, while losing electrons makes the ion smaller than the neutral atom. For example, if an ionic compound is composed of a cation and an anion that has an atomic size which is three times larger, the distance between the two nuclei should be divided by 4 in order to obtain the cation radius. \) shows EA values versus position on the periodic table for the s- and p-block elements.Now, reasonably, we determine the atomic radius OF ANY SPECIES by the radius of its valence electron. An ions size, compared to its parent atom, depends on whether it gains or loses electrons. Ionic size rises with increasing coordination number for the same ion, and an ion in a high-spin state is bigger than an ion in a low-spin state.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
